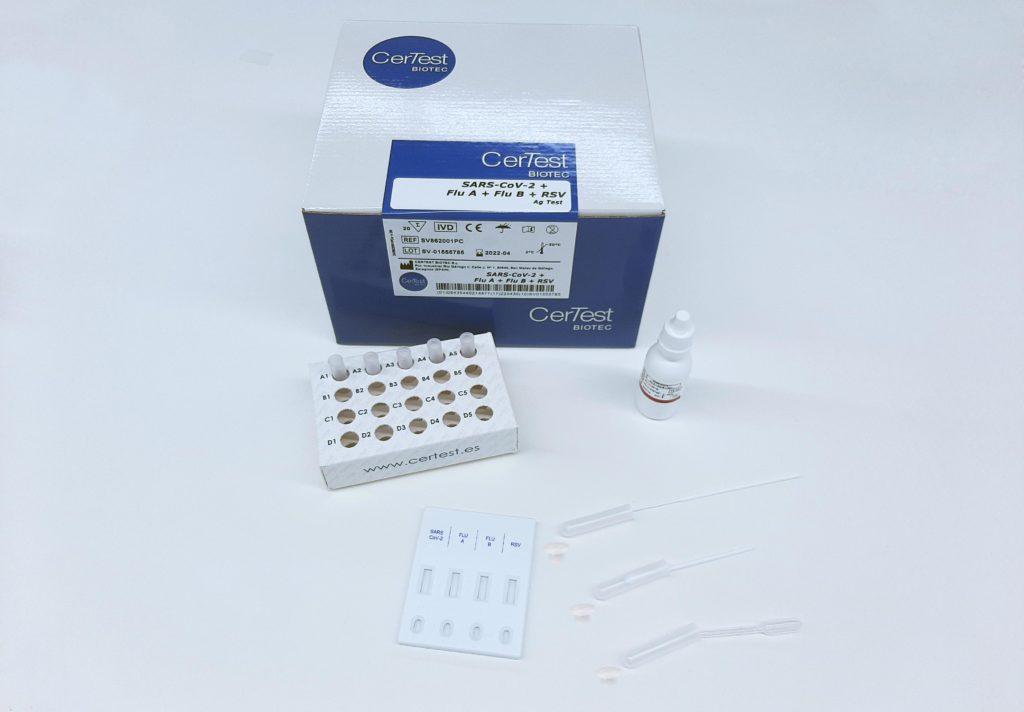
The SARS-CoV-2/Influenza A+B/RSV Antigen Combo Rapid Test is intended for use by trained clinical laboratory personnel. A rapid immunoassay for the detection of respiratory syncytial virus (RSV) antigens in nasal wash, NP aspirate, NP swab and nasal swab samples. An antigen may also form inside the body. An antigen may be a substance from the environment, such as chemicals, bacteria, viruses, or pollen. This means your immune system does not recognize the substance, and is trying to fight it off. 6.Components from difference lots must not be mixed or used together. An antigen is any substance that causes your immune system to produce antibodies against it. 5.Please read all information in this leaflet before performing the test. 4.The test cassette should remain in the sealed pouch until use. 3.Perform the test at room temperature 15 to 30☌. 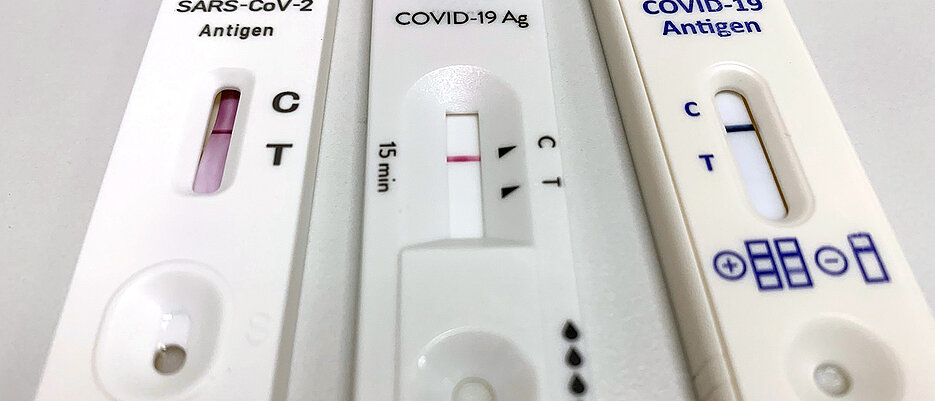
Negative results should be considered in the context of a patient’s recent exposures, history and the presence of clinical signs and symptoms consistent with SARS-CoV-2, Influenza A+B and RSV. Safety Information: 1.For in vitro diagnostic use only. Negative results should be treated as presumptive and confirmed with a molecular assay, if necessary for patient management. Negative results do not preclude SARS-CoV-2/Influenza A+B/RSV infection and should not be used as the sole basis for treatment or patient management decisions. The agent detected may not be the definite cause of disease. rapid, qualitative detection of respiratory syncytial virus (RSV) antigen. The sample is then tested for the presence of viral RNA. Easy to use, fast and accurate, Quidels innovative line of Rapid Visual Tests. Positive results do not rule out other bacterial/viral infection. Sometimes referred to as a PCR test or a nucleic acid amplification test (NAAT), these tests require a sample from the upper respiratory tract, typically a swab obtained from the nose. Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status. An antigen is generally detectable in upper respiratory specimens during the acute phase of infection. Results are for the detection of SARS-CoV-2, Influenza A+B and RSV Antigens. The SARS-CoV-2/Influenza A+B/RSV Antigen Combo Rapid Test (Nasopharyngeal Swab) is a rapid chromatographic immunoassay for the qualitative detection of SARS-CoV-2, Influenza A, Influenza B and Respiratory Syncytial Virus(RSV) antigens in nasopharyngeal swab specimens from individuals with suspected SARS-CoV-2/Influenza/RSV infection in conjunction with clinical presentation and the results of other laboratory tests.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
